
A recently published cohort study with a register-based follow-up of some individuals from the Danish general population sought to investigate whether smokers are at an increased risk for developing MPNs versus those who have never smoked.
New Tool May Provide Deeper Understanding of Glioblastoma Multiforme

A recently published cohort study with a register-based follow-up of some individuals from the Danish general population sought to investigate whether smokers are at an increased risk for developing MPNs versus those who have never smoked.

With its new FDA approval, Hemlibra (emicizumab-kxwh), a bispecific factor IXa- and factor X-directed antibody, has become the first prophylactic treatment for patients with hemophilia A with or without factor VIII inhibitors.

Myeloproliferative neoplasms have varied progression rates, and a complex genetic landscape may contribute to heterogeneity in the outcomes of patients with these diseases.
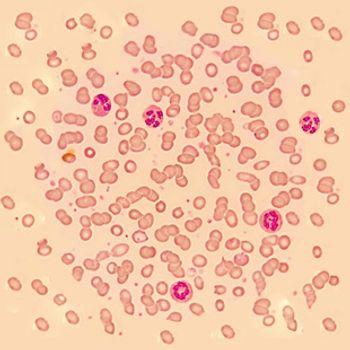
Abnormal concentrations of inflammatory markers detected in children with B-cell precursor acute lymphoblastic leukemia (ALL) support the theory that children with ALL are born with dysregulated immune function.
A recent paper sought to address the knowledge gap in the literature with respect to the effects of cardiovascular events and thromboembolic events on healthcare cost associated with patients with polycythemia vera receiving hydroxyurea.

The ornithine decarboxylase 1 (ODC1) gene, for the first time in humans, has been linked to a new pediatric disorder leading to macrosomia, macrocephaly, developmental delays, alopecia, hypotonia, and hearing loss, according to a new report.
A recent open-label, single-arm, phase 2 study sought to assess evaluate an alternative dosing regimen for adults myelofibrosis (MF) to assess whether such a regimen could mitigate anemia.




The new initiative will not focus on donor-related bone marrow transplants in sickle cell disease, but instead will home in on genetic modifications of a patient’s own stem cells.
The ongoing REVEAL study, a prospective observational study of US patients with the myeloproliferative neoplasm polycythemia vera (PV), is designed to gather data on disease burden, clinical management, patient-reported outcomes (PROs), and healthcare resource utilization of this patient population.

In their study published in Nature Communications, the authors mapped out the epigenomic profiles of rheumatoid arthritis (RA) and osteoarthritis for the first time to provide insight on RA-specific pathways and transcription factor motifs. A surprising discovery was that the Huntingtin-interacting protein-1 may play a role in the pathogenesis of RA.

While the pathogenesis of autoimmune myelofibrosis is not completely understood, in patients with systemic lupus erythematosus, it may be triggered by circulating immune complexes that induce megakaryocytes to release platelet-derived growth factor.

Using whole-exome sequencing and evaluating germline mutations, researchers have discovered novel mutations in several genes among patients with essential thrombocythemia and primary myelofibrosis, which could influence the treatment they receive.
According to treatment recommendations from the European LeukemiaNet and the National Comprehensive Cancer Network, patients with high-risk polycythemia vera (PV) should receive cytoreductive medication together with standard treatments for low-risk PV: phlebotomy and low-dose aspirin.
Patients with neurodegenerative ataxia that are treated with cerebello-spinal transcranial direct current simulation (tDCS) have significant improvements in gait, upper limb coordination, quality of life, and other neurological evaluations of cerebellar deficits, according to results from the CStDCSAtaxia trial.
The COMBI study, a prospective, open‐label, single‐arm phase 2 study ongoing in Denmark, is investigating the feasibility of treating patients with low- to intermediate-risk myelofibrosis or polycythemia vera with low-dose pegylated interferon alfa-2 in combination with ruxolitinib.

Ovid Therapeutics Inc., a biopharmaceutical company, recently announced that the phase 2 STARS trial, which tested the efficacy of the investigational drug OV101, achieved its primary endpoint of safety and tolerability for treating Angelman syndrome.
Extramedullary hematopoiesis—the presence of hematopoietic activity in sites outside of the bone marrow—can occur in malignant and non-malignant hematologic diseases, and they may be a complication of primary myelofibrosis.

