News
Article
Brodalumab Is Effective, Safe in Patients With Moderate to Severe Psoriasis
Author(s):
Posters presented at the American Academy of Dermatology Annual Meeting demonstrated both short- and long-term efficacy and safety profiles of brodalumab in patients with moderate to severe psoriasis.
This content was produced independently by The American Journal of Managed Care® and is not endorsed by the American Academy of Dermatology.
Two posters presented at the American Academy of Dermatology 2024 Annual Meeting show brodalumab to be effective and safe in treating patients with plaque psoriasis in studies conducted at 3 months and up to 3 years after treatment administration.
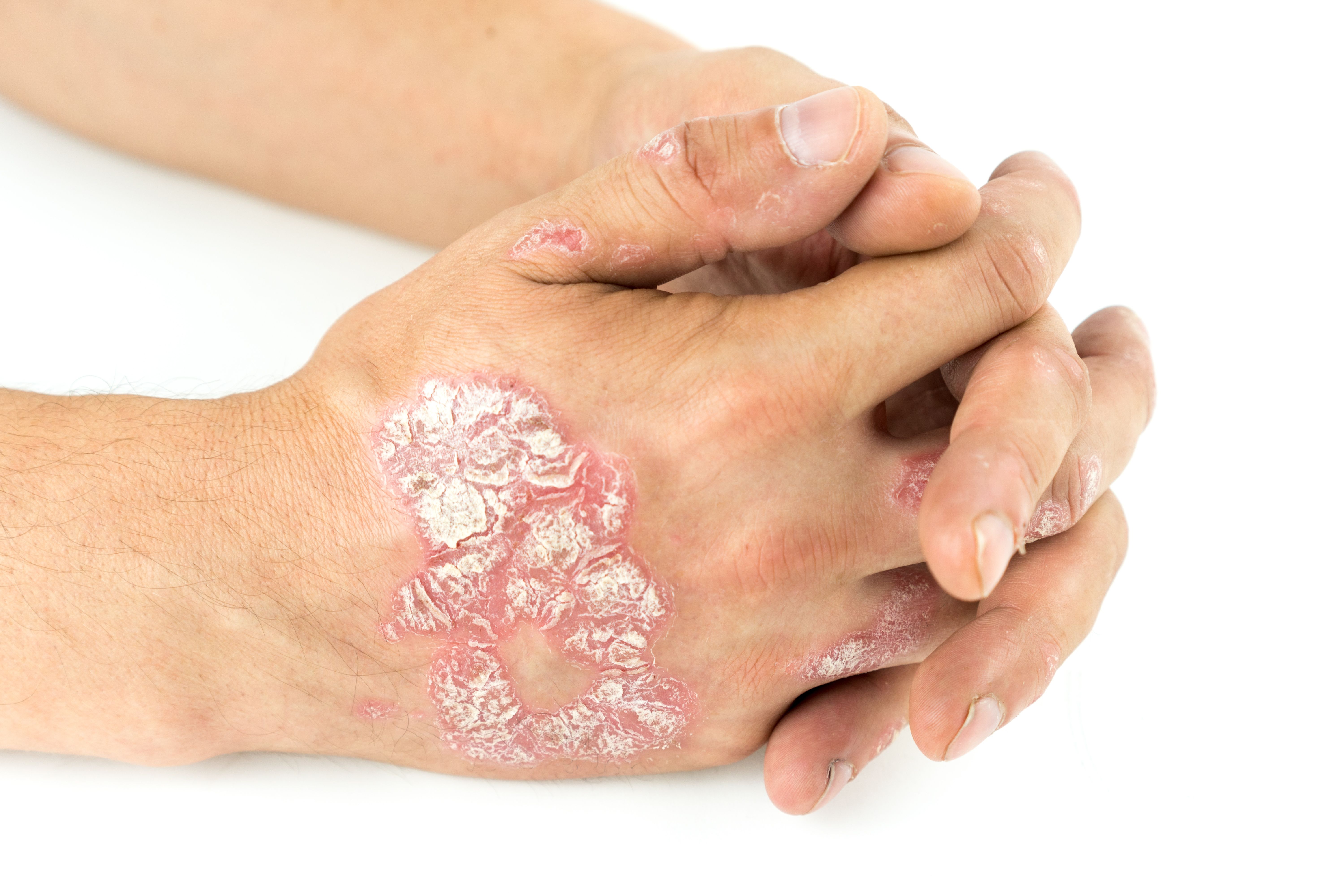
Brodalumab, a monoclonal antibody and interleukin-17 receptor A antagonist, is approved for the treatment of patients with moderate to severe psoriasis. However, real-world data pertaining to predictive factors for long-term survival are limited.
In the first poster, researchers aimed to measure the efficacy and safety of brodalumab up to 3 years in a real-world setting.1 The researchers used medical records from 199 patients with chronic plaque psoriasis treated with brodalumab between December 2019 and 2021 to determine the treatment response rates at weeks 16, 28, 52, 78, 104, and 156, according to Psoriasis Area and Severity Index (PASI) 100, PASI 90, and an absolute PASI 3 or less, as well as long-term drug survival. Additionally, the researchers evaluated possible predictive factors associated with the achievement of response outcomes and possible predictive factors associated with lower drug survival.
At 16 weeks, 80.3% of patients achieved a PASI of 3 or less, 66% achieved PASI 90, and 54.3% achieved complete disease clearance. However, mild loss of response was noted at later time points, particularly for PASI 100 (55.2%) and PASI 90 (65.5%) at week 156. Furthermore, 66.22% of patients continued with therapy after 156 weeks of treatment, with IL-17 appearing to be associated with an increased risk of treatment discontinuation (HR, 2.51; 95% CI, 1.06-5.98; P = .037), and achievement of PASI 3 or less until 16 weeks with less risk (HR, 0.27; 95% CI, 0.14-0.51; P < .001).
Other predictive factors associated with treatment response were bio-naïve status and high body mass index.
The second poster aimed to describe the real-world outcomes of brodalumab for patients with moderate to severe plaque psoriasis in Canada 3 months post initiation.2
The interim analysis was based off a subset of patients who had participated in CARE, an ongoing multicenter, 12-month, prospective, observational study. Eligibility criteria included that the decision to initiate brodalumab therapy must have been reached before and independently of the study recruitment, and before the patient received their first dose. All study visits were conducted at baseline and at 3, 6, and 12 months after treatment initiation, with data collection beginning in October 2021 and continuing until January 2025.
At the 3-month follow up, 63.7% of patients achieved a static Physician’s Global Assessment (sPGA) score of clear (0) or almost clear (1), and most patients (69.6%) achieved a 2-grade or more improvement of the sPGA score compared with baseline.
Additionally, the mean (SD) absolute and percentage change in Psoriasis Symptom Inventory (PSI) were −13.6 (8.7) and −68.6% (35.6%), respectively. Furthermore, 97.9% of patients continued with brodalumab at 3 months post initiation.
The researchers acknowledged some study limitations, such as being observational, relying on data availability in patient charts, and missing patient-reported outcomes. Additionally, the study samples were limited to those who were willing and able to provide patient-reported outcomes data using an online platform.
Furthermore, the researchers noted that all enrolled patients will continue to be monitored for up to 12 months after treatment initiation to provide further information on the real-world outcomes of brodalumab among this patient population.
References
1. Mastorino L, Dapavo P, Burzi L, et al. Drug survival, effectiveness, and safety of broadlumab for moderate to severe psoriasis for up to 3 years. Poster presented at: American Academy of Dermatology Annual Meeting 2024. March 8-12, 2024; San Diego, CA. Poster 51352. https://aad-eposters.s3.amazonaws.com/AM2024/poster/51352/Drug+survival+effectiveness+and+safety+of+brodalumab+for+moderate-to-severe+psoriasis+up+to+3+years.pdf
2. Gaudet V, Wiseman M, Kalia S, et al. Effectiveness and persistence of brodalumab in patients with plaque psoriasis in the Canadian real-world setting: 3-month follow-up results from the CARE study. Poster presented at: American Academy of Dermatology Annual Meeting 2024. March 8-12, 2024; San Diego, CA. Poster 49513. https://aad-eposters.s3.amazonaws.com/AM2024/poster/49513/Effectiveness+and+Persistence+of+Brodalumab+in+Patients+with+Plaque+Psoriasis+in+the+Canadian+Real-World+Setting+3-Month+Follow-up+Results+from+the+CARE+Study.pdf