Hematology
Latest News
Latest Videos

More News
X. Long Zheng, MD, PhD, explains when physicians should administer caplacizumab after suspecting a diagnosis of acquired TTP, and discusses the risk of relapse or refractory disease.

An expert in acquired TTP describes the use of caplacizumab in treating acquired TTP, and highlights data from a phase III clinical trial.
X. Long Zheng, MD, PhD, details the typical healthcare costs for acquired TTP, the treatment options available to patients, and the goals of the healthcare provider.
A key opinion leader explains how acquired TTP is diagnosed, with a focus on the use of ADAMTS13 testing.

X. Long Zheng, MD, PhD, gives an overview on acquired thrombotic thrombocytopenic purpura including prevalence and the typical patient presentation.
The CDC's Advisory Committee on Immunization Practices voted to uphold the original language of the emergency use authorization for Johnson & Johnson's COVID-19 vaccination, and is possible the 1-dose vaccine could resume Saturday.

The in vitro study of melanoma cells analyzed the molecular mechanisms of the pair of proteasome inhibitors, finding that they reduced B16-F1 tumor growth.

After cases of rare and severe blood clots led to 1 death and 1 hospitalization, the CDC and FDA will pause the use of Johnson & Johnson's (J&J) COVID-19 vaccine.

Patients who experienced a fall within 12 months of multiple myeloma (MM) treatment initiation had a shorter median survival compared to those without falls.

Multiple myeloma is the second most common blood cancer, affecting more than 130,000 US patients.
Disease severity was strongly associated with health-related quality of life, moderately associated with use of disability insurance, and weakly associated with household income for patients with sickle cell disease.

Updates to the NCCN Guidelines in multiple myeloma reflect recent trial results, giving clinicians and patients many choices.

The safety and effectiveness of the long-acting agent is backed by data from trials of the drug among treatment-experienced individuals living with HIV-1 whose viral load is on the rise due to other regimens failing. Investigation continues among these patients, as well as treatment-naive patients.
Researchers describe the case of an older patient who underwent myeloma drug sensitivity testing as part of an effort to identify the therapies most likely to produce a response in this difficult-to-treat population.
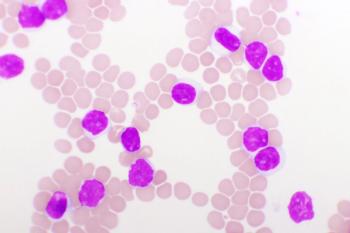
Patients with multiple myeloma (MM) often relapse over time despite initially achieving a complete response without minimal residual disease following induction treatment, leaving questions about changes in the immune system and the prognosis of the disease.
More than 2 years after treatment, some of the patients with relapsed/refractory multiple myeloma have yet to see a relapse. An FDA decision on the therapy is expected within a month.

The peptide-drug conjugate rapidly delivers an alkylating payload into tumor cells.

Health-related quality of life was maintained when treating relapsed/refractory multiple myeloma (RRMM) with daratumumab in combination with bortezomib and dexamethasone.
Sustained minimal residual disease (MRD) negativity may predict long-term outcomes in relapsed/refractory multiple myeloma (RRMM), and daratumumab-based combinations show higher rates of sustained MRD negativity compared with the standard of care.
Carfilzomib-dexamethasone improved renal overall and complete response in patients with relapsed or refractory multiple myeloma (RRMM) compared with a regimen of bortezomib-dexamethasone in a real-world study.

Whereas patient care was often bogged down in bureaucracy when he was younger, Robert K. Massie Jr, PhD, MA, of the Society for Progress, notes that hospitals and caregivers are increasingly paying attention to what makes sense for patient care.

The FDA issues an emergency use authorization (EUA) for Eli Lilly’s coronavirus disease 2019 (COVID-19) treatment; insurance waivers for COVID-19–related costs are starting to expire; new data update convalescent plasma’s indication.
Experts in the management of multiple myeloma consider promising developments for the future of relapsed/refractory treatment, with special consideration to bispecific antibodies and CAR (chimeric antigen receptor) T-cell therapy.

Patients’ socioeconomic status is a significant factor in predicting survival rates for multiple myeloma, according to a new study.

Key opinion leaders consider the role of venetoclax in managing patients with relapsed/refractory multiple myeloma.